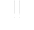Darlene M. Anthony (1) , Chris Groves (1) and Joe Meiman (2)
(1) Center for Cave and Karst Studies, Department of Geography and Geology,
Western Kentucky University, Bowling Green, KY 42101, USA
(2) Division of Science and Resource Management,
Mammoth Cave National Park , Mammoth Cave , KY 42259 , USA
Abstract
Many geochemical studies have been made of karst waters worldwide. Most data that provide the framework for our current understanding of the evolution of karst waters have come from sampling at discrete times and locations, such as springs or wells. Relatively few studies have been made of the geochemical evolution of groundwater as it moves through an open flow system. This paper addresses the seasonal changes in the geochemistry of the Logsdon River conduit as it passes through nearly 10km of the carbonate aquifer of south-central Kentucky .
The most important control on the ability of groundwaters to dissolve limestone is their carbon dioxide pressure, which is influenced by a variety of complex interactions with soil, bedrock, and in-cave organic decay. The fieldwork involved in this research combines seasonal sampling of the entire traversable length of the Logsdon River conduit, as well as continuous monitoring of the chemistry at key points within the flow system. Preliminary results of this study indicate both seasonal changes in CO 2 , transport through the Mammoth Cave karst aquifer during summer and winter conditions, along with significant geochemical changes as the water moves through a distance of 10km.
Keywords: Chemical evolution of groundwater, hydrochemical monitoring, karst hydrochemistry, Mammoth Cave
Introduction and background studies
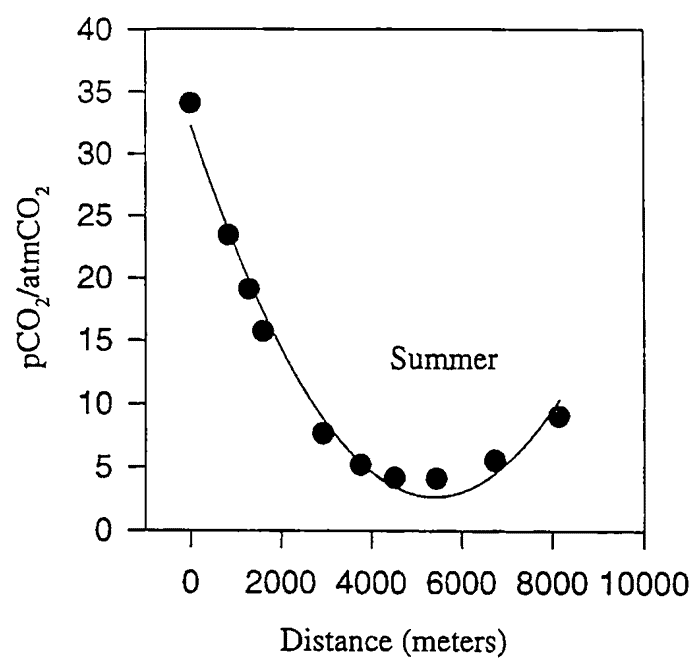
The Logsdon River within the Mammoth Cave System drains the Sinkhole Plain of Central Kentucky between the towns of Park City and Cave City to its discharge spring at Turnhole Bend on the Green River , a distance of nearly 23 kilometers (Fig. 1). It is possible to follow the Logsdon River through nearly 10km of cave passage before diving equipment is needed to continue at either end, making the Logsdon River one of the world's longest continuously-traversable underground rivers. This allows for the investigation of changes in the geochemistry of the groundwater as it moves through nearly 10 kilometers of open conduit flow. [Note: The Logsdon River was discovered in 1979 at two separate locations in the Mammoth Cave System by independent parties of explorers. It was named Hawkins River by those who had found its downstream end in Proctor Cave , and Logsdon River in the upstream reaches where it was found in Morrison Cave , and later in Roppel Cave . The downstream end, which is formed by the confluence of a right-and left-hand branch, is referred to in the literature as the Right and Left Forks of the Hawkins River . The Right Fork drains the Park City, KY area, and is traversable for only a few hundred meters upstream before water completely fills the passage. The Left Fork was connected upstream with the Logsdon River in Morrison and Roppel Caves , and drains the Cave City, KY area. It is this portion of the conduit that may be traveled for a long distance, and is the focus of this study.]
In one of the first hydrochemical studies of the Central Kentucky karst aquifer, more than 80% of the runoff collected on the Sinkhole Plain recharge area was found to be discharged at regional springs on the Green River (Hess, 1974). Hess concluded that the majority of groundwater passing through the aquifer was being carried by a conduit system. At that time, access to the Tumhole Bend conduit system was limited to sinking streams, karst windows at Cedar Sink ( Owl Cave ) and Mill Hole, and Turnhole Spring itself. The horizontal component predicted to exist between the Sinkhole Plain and Turnhole Spring was inaccessible until the 1979-83 discoveries and connections of caves containing pieces of the Logsdon River .
Fig. 1. Location of the Logsdon River within the Turnhole Spring groundwater basin (after Meiman and Ryan, 1993).
It is not often that a baseflow conduit can be physically followed for as great a distance as the Logsdon River . Typically, open conduits end quickly in sumps or siphons, explorable only by cave divers. Sumps represent the end of the open system, as the water is no longer in contact with a cave atmosphere. For this reason, relatively few studies have been made of the geochemical evolution of karst waters within the actual aquifer (Groves, 1992; Wicks and Englen, 1996). The accessible portion of the Logsdon River conduit begins on the topographic boundary between the Sinkhole Plain and the Mammoth Cave Plateau, and provides access to one of the longest (and arguably, one of the most spectacular) sections of conduit flow in the world. Geochemical data from the Logsdon River conduit gathered during this study detail the evolution of groundwater chemistry within the Turnhole Spring basin. Combined with the data from prior studies, a more complete understanding of the geochemical evolution of a mature karst flow system is made.
In the early 1980's, two sets of wells were drilled into each of the Forks of the Hawkins River . The primary purpose was to monitor the effects of storm and seasonal changes on the water quality of these two streams, and to evaluate the impact of contaminants coming into the groundwaters of Mammoth Cave National Park (MCNP) from sources outside park boundaries. To facilitate maintenance and upgrades at the wells, a 30m-deep shaft was also drilled beneath Doyel Valley , considerably shortening travel time to the welts. Continuous monitoring of the temperature, specific conductance, pH, and stage height, as well as water analyses at the Left and Right Forks yielded surprising information; the groundwaters were, at least part of the time, oversaturated with respect to calcite (Groves and Meiman, 1996). The saturation index for calcite (SI cal ), the primary mineral in limestone, is a useful indicator of the ability of the groundwater to dissolve limestone. It is an empirical number defined as the ratio of the ion activity product to the solubility product constant (White, 1988). Used to describe the interaction between groundwater and the cave walls, negative values indicate that the water is undersaturated with respect to calcite and thus will dissolve it; positive values indicate that the water is saturated, potentially precipitating rimstone or flowstone. Sinking streams on the Sinkhole Plain, local springs and karst windows on the Mammoth Cave Plateau, and regional springs on the Green River have been found to be undersaturated with respect to calcite (Hess, 1974; Hess and White, 1989). With the addition of new data from the Left Fork well, it was concluded that the rate of cave development within the Logsdon River conduit was not continuous (Groves and Meiman, 1996).
Data from the Left Fork well provided a window into what was happening in the aquifer between the recharge area and Turnhole Spring. However, it was still not clear at what point in the conduit the water changes from an undersaturated state to a saturated state at the well. Therefore, the next logical step in understanding the limestone-groundwater interaction in the aquifer was to examine the geochemistry in more detail between the Left Fork well and the farthest upstream end of the Logsdon River . At some point, there would be an exact location in the conduit where the transition from an undersaturated condition to a saturated condition is made. To this end, a pilot study of the changes in the geochemistry of the Logsdon River from the Left Fork well to the farthest upstream sump in Roppel Cave was proposed as a preliminary investigation for a master's thesis, and conducted during the 1996-97 school year during two seasonal extremes in the karst aquifer.
Pilot study procedures
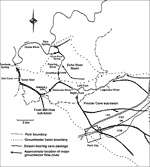
In order to collect data and sample undisturbed water along the entire length of the conduit, the pilot trip began at the Left Fork well and moved upstream to the S188 sump in the Roppel Cave section of the Mammoth Cave System. In the pilot study, ten sampling sites in the Logsdon River were selected for even distribution of the total distance involved, and for significant geologic and/or hydrologic features encountered en route (Fig.2). The first sample was drawn from the Left Fork well, and the last sample was taken from the S188 sump in the Roppel Cave section. At each site, the following measurements or samples were taken:
a) temperature
b) pH
c) conductivity
d) 500mL water sample
Fig. 2. Sampling sites in the Logsdon River pilot study.
Temperature, pH, and conductivity probes were carried in waterproof cases. At each sampling site, the probes were placed upstream of the party in undisturbed flowing water, and allowed to stabilize before readings were recorded. After the probes had been removed from the water, a 500mL sample of water was taken from undisturbed water. The sampling party then moved to the next site .
Standard procedures for collecting water samples were followed. Sterile bottles were rinsed three times in the water to be sampled. The bottles were then filled and tightly capped underwater, after all air bubbles were shaken out. Great care was exercised in moving the sample bottles through the cave, to avoid smashing or puncturing the bottles. Once brought to the surface, the bottles were placed in a cooler and kept on ice until received by the Ogden Environmental Laboratory at Western Kentucky University for analysis.
The water samples removed from the Logsdon River were analyzed for calcium, magnesium, and bicarbonate by the Ogden Environmental Laboratory at Western Kentucky University . These data, along with the other variables measured in the cave, were used to calculate the geochemical parameters which directly describe the saturation state of calcite and the partial pressure of carbon dioxide. All data were stored and manipulated using Sigmaplot© software. Regression analyses of the changes in geochemical variables and parameters over distance were done, with distance from the upstream S188 sump used as the independent variable.
The first sampling trip was done on October 12-13, 1996 , which represented low-flow summer conditions in the aquifer. The second sampling trip was done on February 22-23, 1997 , which represented winter conditions in the aquifer. Although the February trip was not completed in its entirety due to an unexpected sump in Roppel Cave , the data gathered was considered sufficient. In analyzing the preliminary data from the pilot study, changes in the carbon dioxide content and the calcium saturation index were found.
Preliminary results (and more questions)
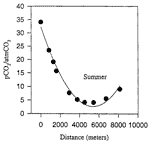
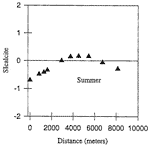
Preliminary analyses of dissolved solids, calcite saturation state, and carbon dioxide pressure of water samples from the summer trip produced the most detailed spatial data to date of the flow system draining the Turnhole Spring basin. Relatively high CO 2 pressures (greater than 35 times that of atmospheric CO 2 pressure) were present at the upstream sampling site (Fig.3). These high CO 2 pressures were perfectly mirrored by the undersaturated state of calcite, indicating a chemically aggressive water at the upstream sampling site capable of dissolving limestone. As the water moved through the conduit, CO 2 was lost in such quantities that the saturation index for calcite was shifted from an undersaturated to a saturated state (Fig.4). At that point in time, and at those locations in the conduit, the Logsdon River was not capable of dissolving limestone. Instead, precipitation of calcite into secondary calcite formations, such as rimstone and flowstone, was possible. Outgassing of CO 2 into the cave atmosphere appears to be the major process controlling the evolution of the groundwater, resulting in the undersaturated waters of the upstream Logsdon River becoming saturated with respect to calcite as transport occurs. Following the Logsdon River downstream, the CO 2 pressure began to increase, again mirrored by the saturation state of calcite. As the CO 2 pressure increased, the saturation index of calcite moved from a saturated to an undersaturated state. Once again, the Logsdon River was chemically able to dissolve limestone in the aquifer.
Fig. 3. Evolution of CO 2 pressure in the Logsdon River during summer conditions.
Fig. 4. Evolution of calcite saturation index in the Logsdon River during summer conditions.
Several questions were not readily answerable after the first sampling trip. What is the dominant source for the CO 2 in the upstream end of the aquifer? How does the amount of CO 2 in the summer water samples compare with the CO 2 present in the groundwater during the winter months, when the soils are biologically inactive (Rossi, 1974; White, 1977)? The source(s) for carbon dioxide in the Logsdon River conduit include soils in the recharge area of the Sinkhole Plain, soils above the conduit on the Mammoth Cave Plateau, and decay of organic material in the conduit itself. Measuring the CO 2 content of the groundwater during the winter months, when bacterial action in the soils is at its lowest, would be an indirect measure of the CO 2 input from decaying organic materials that accumulate in the conduit. Comparing this with measurements of the CO 2 content during the summer months, when bacterial action in the soils is highest, would allow investigators to interpret the dominance of CO 2 input from outside sources, such as soils, or in-cave sources, such as decay of organic materials and other biological activities in the Logsdon River .
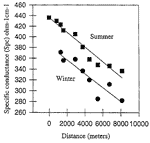
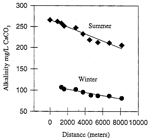
The winter sampling trip was plagued by a malfunctioning pH meter and an unexpected turnaround at a sump, just two sites short of exiting the Logsdon River in Roppel Cave . Data for CO 2 pressure and SI cal was therefore limited to the first eight sampling sites. Incomplete as it was (compared with the summer sampling trip), the data represented the most complete spatial sampling of the geochemistry in the Logsdon River conduit during winter conditions to date. Initial comparisons of chemical data between summer and winter samples were made using regression models (Figs 5 and 6).
Discussion
As groundwater moves through the Logsdon River conduit, changes in the geochemistry occur. The geochemical evolution of the Logsdon River is affected by seasonal extremes in the aquifer, as well as by the distances through which the water is transported during both summer and winter.
Some of the more striking changes in the geochemistry involve the interaction between carbon dioxide pressures and calcite dissolution/precipitation. The high amount of carbon dioxide present during the summer season at the upstream end of the Logsdon River was a surprise, even though the leaves had not fallen from the trees, and the soil was still warm and quite biologically active. A sharp decrease in the CO 2 pressure occurred over the first 4000m, due to outgassing of CO 2 between the cave atmosphere and the CO 2 -rich water. This created a saturated condition with respect to dissolved calcite, which may be precipitated as secondary calcite in the form of rimstone dams and flowstone. Anyone who has made the trip into the Logsdon River will recall portions of the conduit filled with spectacular rimstone dams and pools, huge meters-high flowstone formations, and other proofs of saturated conditions. During the summer sampling trip, these saturated conditions did not remain constant for long. The CO 2 levels in the water began to increase, driving the groundwater chemistry back towards the undersaturated, or dissolving state with respect to calcite. The source of the additional carbon dioxide found at 5000m is unknown at this time. Possible explanations include the mixing of a tributary (infeeder) with a different chemical signature into the Logsdon River or the addition of carbon dioxide by in-cave organic decay.
Although the winter sampling trip did not yield the quantity of data as did the summer trip, seasonal differences were apparent when comparing the measurements of dissolved calcium, bicarbonate, and specific conductance. Changes in these measurements also occurred over the entire transport distance.
Future fieldwork
Additional sampling trips over the entire distance of the Logsdon River conduit during seasonal extremes will be conducted during the summer of 1997 and the winter of 1998. The sampling trips will be made during summer (low-flow) conditions and winter conditions. All sampling trips will follow the procedures outlined above. Precise hydrologic and geochemical conditions will be closely checked at the Left Fork well 24 hours before a trip is scheduled.
An automatic data recorder will be constructed, consisting of temperature, conductivity, pH, and stage height probes, to be installed at the S188 sump in the Logsdon River during the summer of 1997. Periodic trips to the sump will be made to download the data, calibrate the probes, and collect water samples for analysis of bicarbonate, calcium, and magnesium. Data will be collected until the spring of 1998. Seasonal changes in dissolved calcium, alkalinity, and CO 2 pressure will be analyzed for statistical significance. Data from this upstream end will be compared with the hydrologic and chemical conditions for the same time periods at the downstream Left Fork well. At the conclusion of this study, comparative analyses of seasonal data will be made, in addition to the examination of spatial changes in each sampling trip. The data collected over the entire investigation will be analyzed and interpreted for statistical and geochemical significance. Research questions will be evaluated based on the data gathered from this study, advancing our understanding of the behavior of the Mammoth Cave karst aquifer in particular, and karst processes in general.
Fig. 5. Seasonal evolution of specific conductance in the Logsdon River.
Fig. 6. Seasonal evolution of alkalinity in the Logsdon River.
Acknowledgements
I wish to thank my advisors, Dr. Chris Groves and Joe Meiman, for support and encouragement beyond belief. Thanks to my summer sampling crew (Phil Bodanza, Sheri Engler, Chris Groves, Dick Market, and Joe Meiman); my winter sampling crew, who survived the two-way Logsdon River Death March (Sheri Engler, Michael Hennion, and Bart Bledsoe); my two children, Thomas and Kathy Borden, who have sacrificed so much for this thing called higher education; to the late Dr. Jim Quinlan, who graciously allowed me to paint stilling wells (who knew then?); and finally, to the dozen or so Logsdon River explorers and surveyors whom I have been so privileged to work with all these years. You know who you are! If you close your eyes, you can hear Roberta laughing!
References
- Groves C. 1992. Geochemical and kinetic evolution of a karst flow system: Laurel Creek, West Virginia. Ground Water 30, 186-191.
- Groves C. and Meiman J. 1996. Speleogenesis of Mammoth Cave : What are we learning at Hawkins River ? Proc. of the Fifth Mammoth Cave Science Conf., Mammoth Cave , KY.
- Hess J. 1974. Hydrochemical Investigations of the Central Kentucky Karst Aquifer System. Ph.D. thesis, The Pennsylvania State University.
- Hess J. W. and White W.B. 1989. Chemical hydrology. In: White W.B and White E.L. (Eds.), Karst Hydrology Concepts from the Mammoth Cave Area. New York : Van Nostrand Reinhold, 145-174.
- Rossi M. 1974. Sur une series de mesures de teneurs en CO 2 de sols tropicaux. Bull. Assoc. Geogr. France 57, 141-144.
- White W.B. 1977. Role of solution kinetics in the development of karst aquifers. Internat. Assoc. Hydrogeol. Memoir 12, 503-517.
- White W.B. 1988. Geomorphology and hydrology of karst terrains. New York : Oxford University Press, 464 p.
- Wicks C. and Englen J. 1996. (in press) Geochemical evolution of a karst stream, Devil's Icebox Cave , Missouri , accepted for publication by Jour. Hydrol.